Take a closer look at Nick Lucid’s energy concept tree from the Science Asylum's What Exactly Is Temperature? and What is Energy? videos. Looking at the concept tree, we can see why temperature is not the same as thermal energy.
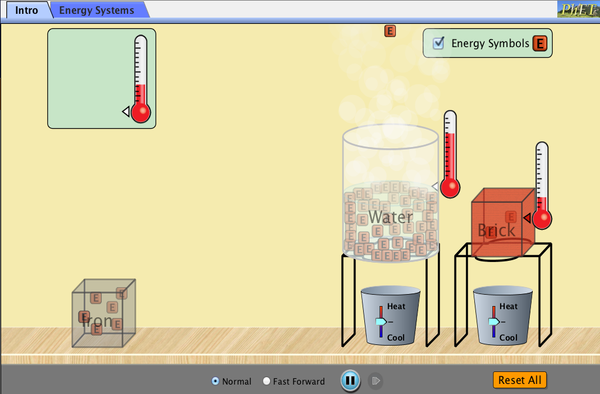
Temperature is the average kinetic energy of the molecule in a substance whereas thermal energy is the total kinetic energy plus the potential energy of the molecules. The thermal energy in a particular substance depends on three things:
- Temperature of an object
- Amount of molecules in the substance (mass)
- Composition of the substance (including state of matter)
So, what about heat? Heat is just thermal energy on the move. Heat flows between substances of different temperatures. Objects have thermal energy, not heat. Objects do, however, transfer their thermal energy through heat. When heat is added to a system, the thermal energy of the molecules increases. When heat is released from a system, the thermal energy decreases. The temperature of the substance also increases or decreases accordingly unless the substance is undergoing a change in state. When substances change state, the potential energy between molecules changes, not the kinetic energy of the molecules.
Review your answer to this question from the thermal energy and temperature exploration: Why do you think that the amount of energy is unequal when the temperatures are equal? Make changes or additions to your answer to reflect your new learning. Compare your answer to the sample answer in the next section, Explain: Debrief